Safety Recall: Medtronic Pain Pumps Have Been Recalled Due For Safety Reasons
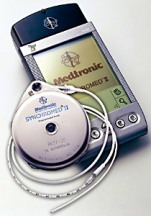 The Medtronic infusion pain pumps are devices implanted in the abdomen of patients to gradually deliver drugs and fluids. These pumps have been known to allow too much of the drugs to be injected and cause overdoses and deaths. These incidents usually involve the refill process. In January 2011, Medtronic sent letters to doctors reminding them of the refill procedures and it has updated its instructions and warning labels.
The Medtronic infusion pain pumps are devices implanted in the abdomen of patients to gradually deliver drugs and fluids. These pumps have been known to allow too much of the drugs to be injected and cause overdoses and deaths. These incidents usually involve the refill process. In January 2011, Medtronic sent letters to doctors reminding them of the refill procedures and it has updated its instructions and warning labels.
Even in light of the extra precautions taken by Medtronic, the FDA (U.S. Food & Drug Administration, last month, issued a Class I recall on Medtronic infusion pumps. Class I recalls are the most serious issued by the FDA/U.S. Government. If you or a loved one has a Medtronic infusion pump you should be aware of the dangers involved.
For more information on the Medtronic infusion pumps, the FDA recall, and your legal rights related to this matter, contact the Ackerson Law Offices and Kentucky recall attorney Brent T. Ackerson at (502) 882-5176 or by clicking here. Know your legal rights and legal options with regards to recalled products.
 Kentucky Personal Injury Attorney Blog
Kentucky Personal Injury Attorney Blog